Research Agreements
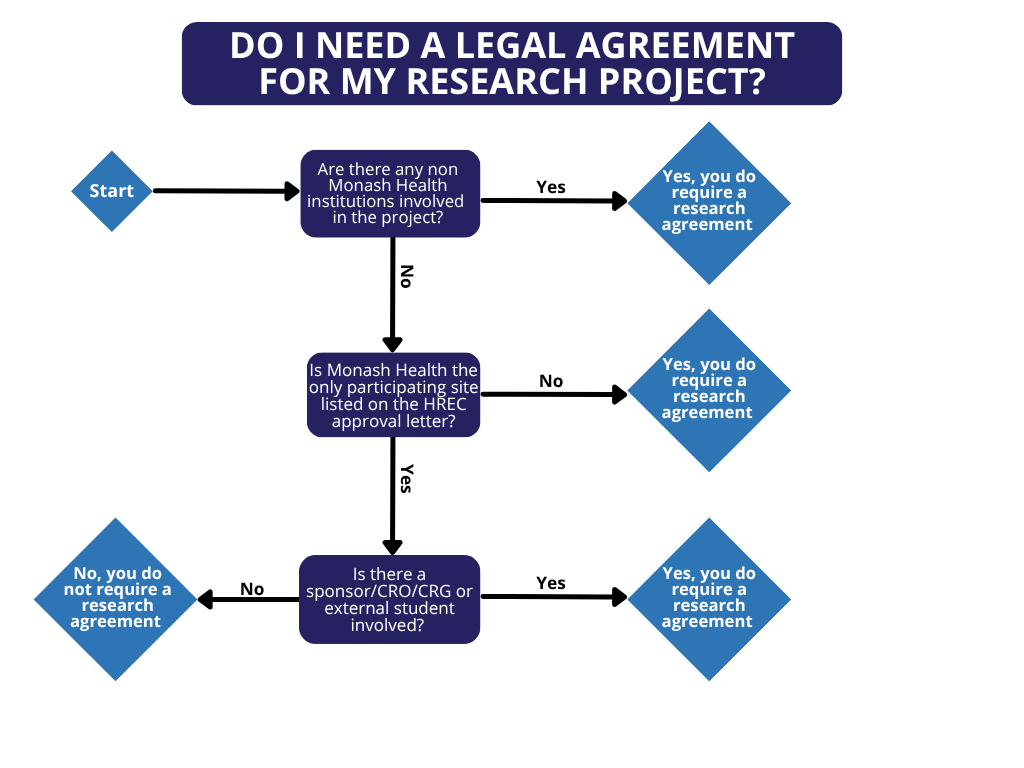
The Monash Health Research Agreements Procedure provide guidance on which agreement to use in which circumstance and the pathway to follow for seeking review and approval.
The preferred templates for collaborative or investigator initiated studies or other circumstances are available in Essential Forms, Templates and Resources.
For all agreements and indemnities, Monash Health is listed as ‘Monash Health (ABN 82 142 080 338), 246 Clayton Road, Clayton, Victoria, 3168.’
Common queries:
Request for Agreement Pre-review
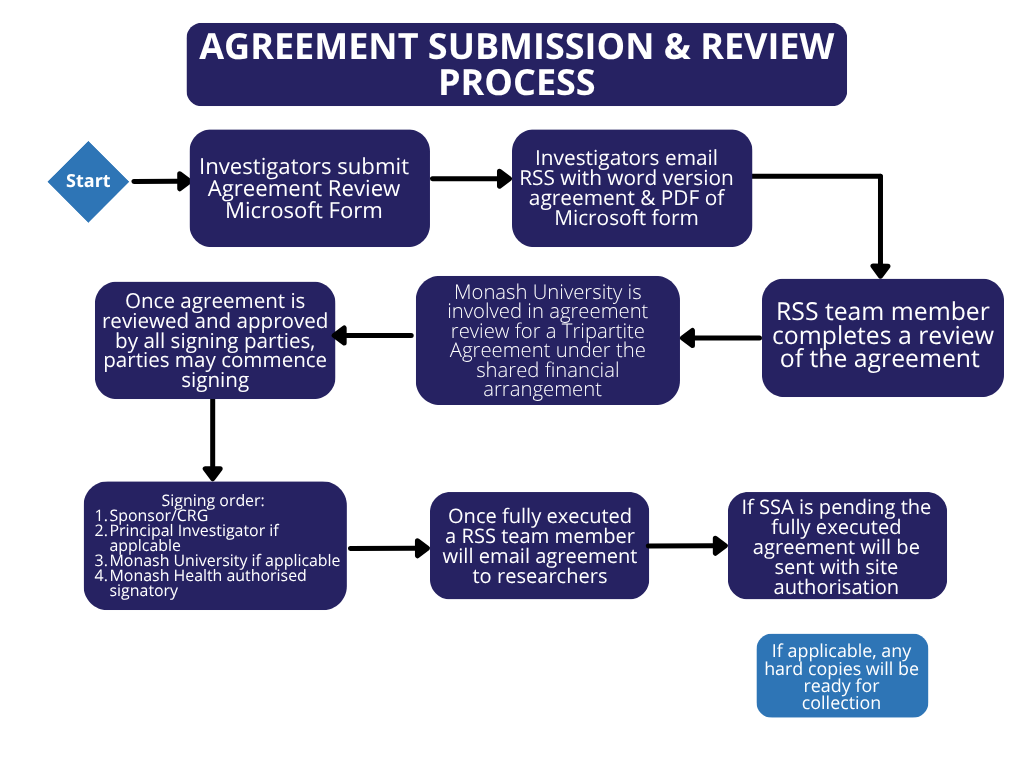
If you have a research agreement and wish to have it pre-reviewed, please submit a Research Agreement Review Request form and email, a word version of the proposed agreement to research@monashhealth.org. Both the form and the agreement submission are required for us to facilitate an efficient legal review process. In your email, please include the Study Title and the Monash Health Local Reference Number (if known) and clarify whether the study has been submitted for Human Research Ethics Review or Site Authorisation.
Agreements for Commercially Sponsored Studies
Monash Health has partnered with Monash University to act as its funds administrator for all commercially sponsored studies except Phase 4 Clinical Trials.
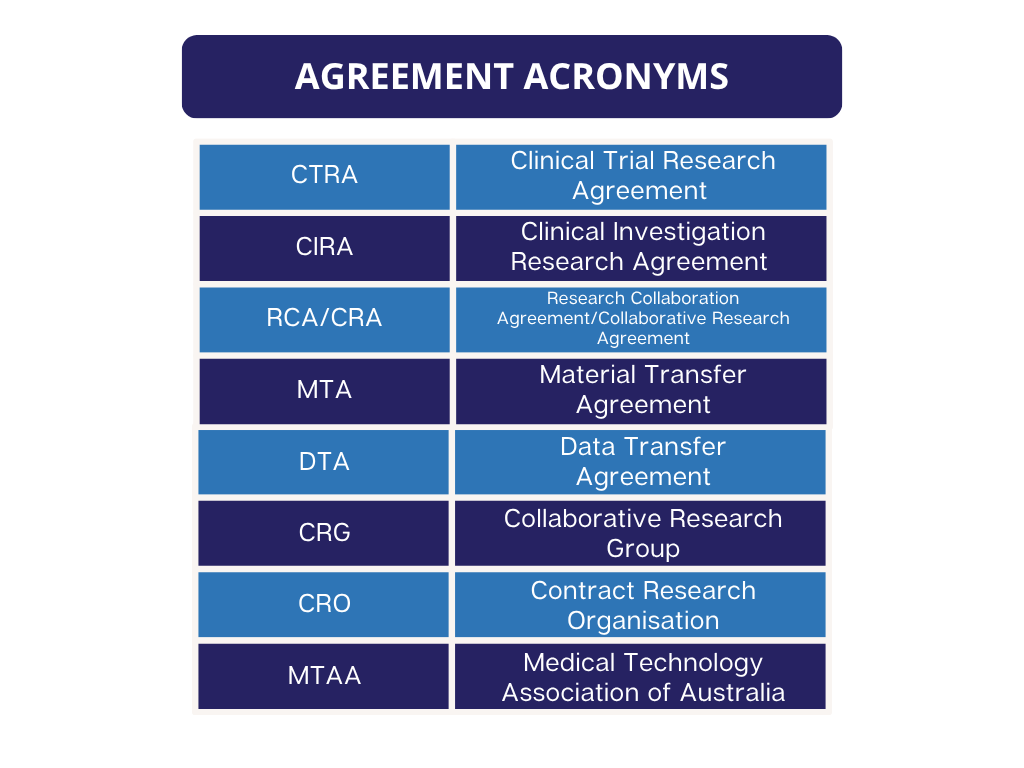
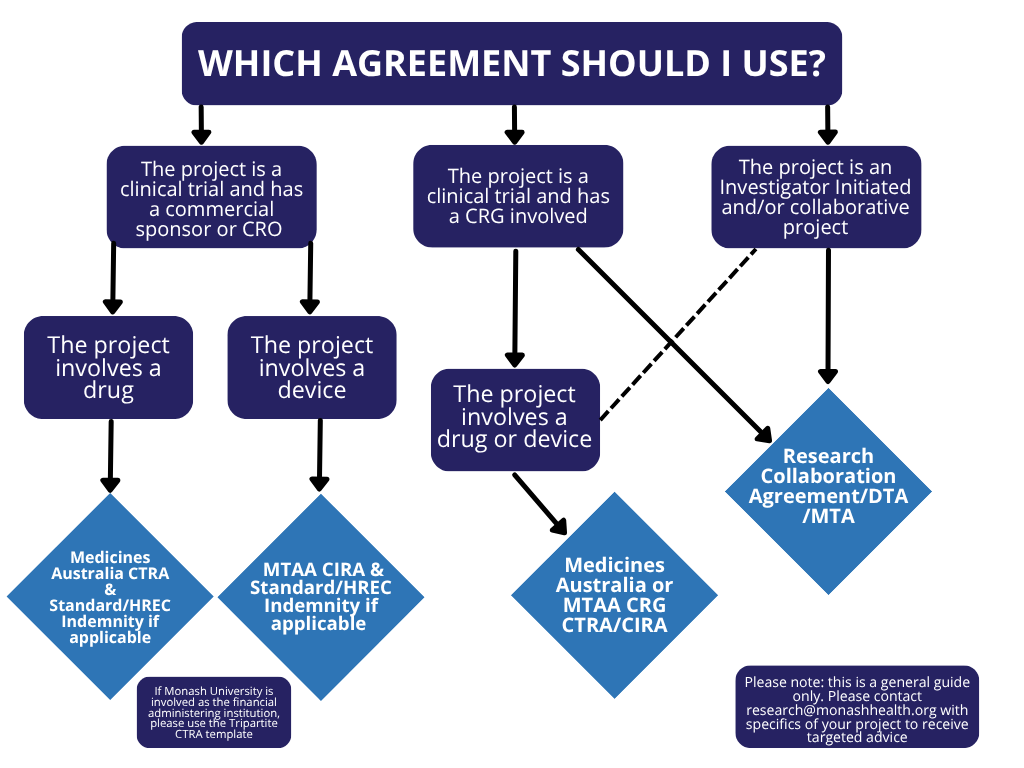
The modified Tripartite Medicine’s Australia agreement templates are for use with commercially sponsored drug trials and the Medical Technology Association of Australia (MTAA) agreement templates are for commercially sponsored device trials.
The Standard Medicines Australia or MTAA templates are to be used for Phase 4 Clinical Trials.
For Standard Indemnities or HREC Indemnities, the Medicine’s Australia and the MTAA indemnity templates are to be used.
Access to Agreement Templates:
1. Tripartite Agreements: Please email research@monashhealth.org to request our Tripartite Agreement templates.
2. Other Agreements and Indemnities: Visit our Essential Forms, Templates and Resources for all other Agreement and Indemnity templates.
Agreements for Collaborative Studies Involving Monash University
The Victorian Research Translation Centres Research Collaboration Agreement template is the preferred agreement. Draft agreements should be emailed to research@monashhealth.org for initial review. Once reviewed by Monash Health, review by Monash University will be arranged through the School of Clinical Sciences.
Research Agreements Training
Our Research Agreement Training session delves deeper into the legal and clinical aspects of research agreements, including:
- Agreement Types
- How to use Agreement Templates
- Agreements and Agreement Variations
- Non Standard Agreements
- Financial Aspects
- Legal Documents: Insurance and Indemnities
Invoicing Agreements (Monash Health access only)
Can’t find what you’re looking for? Contact us.


